SAP Advanced Track and Trace for Pharmaceuticals: Driving the Mission to Vaccinate the World
SAP ATTP Enables COVID Vaccine Manufacturers to Overcome Supply Chain Vulnerabilities. Find How!
Vaccinating the whole world and making everyone resistant to COVID-19 is a daunting task. From managing product life cycles to supply chain tracking, and massive distribution, SAP Advanced Track and Trace for Pharmaceuticals (SAP ATTP) is helping the world’s leading vaccine manufacturers.
With regulatory requirements looming, meeting the target of delivering COVID vaccines to millions will require an efficient system that will eliminate any hiccups in the supply chain. Pharmaceutical companies must examine serialization and track and trace solutions to control the chain of custody to prevent tampering, counterfeiting, theft, and diversion.
Read more: 7 Practical Steps to be Implemented by Organizations that Engage in COVID-19 Vaccine Delivery
Thankfully, SAP ATTP (SAP Advanced Track and Trace for Pharmaceuticals) has emerged as a solution. It can enable your company to comply with serialization and associate reporting requirements. As SAP ATTP was co-developed with the leading pharmaceutical companies, it can be tailored to address the specific needs of your pharma company and your stakeholders. This blog will explain how SAP ATTP can quicken and smoothen the race to vaccinate the world. Let us begin by understanding what SAP ATTP is.
What is SAP ATTP?
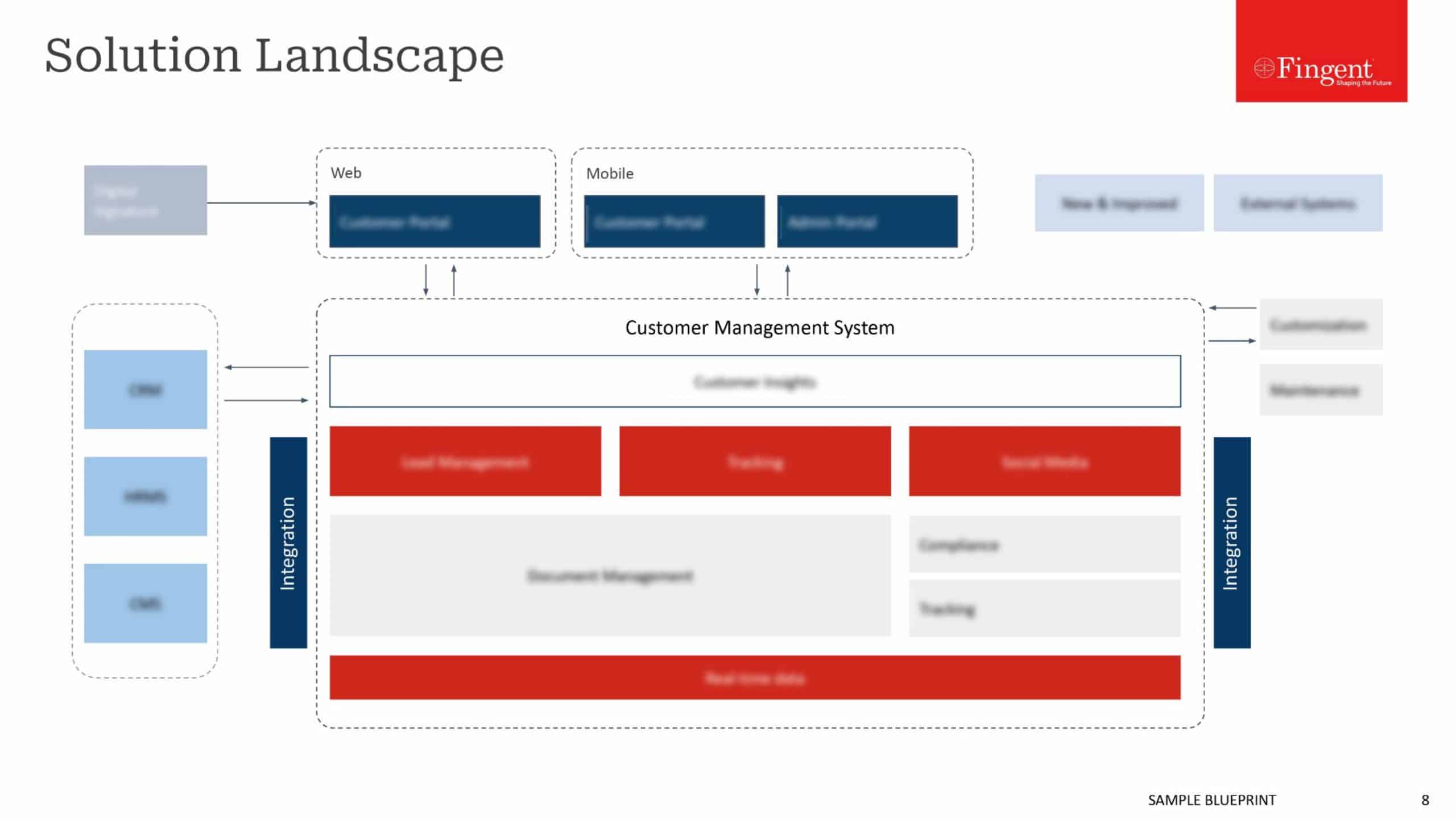
As the name SAP Advanced Track and Trace for Pharmaceuticals suggests, SAP ATTP involves the journey of the vaccine from its source (the pharmaceutical company) to the destination (the end-user) within the supply chain. SAP ATTP enables companies to enforce specific measures to identify the product and view its journey to reduce counterfeit and stolen vaccines.
It addresses the regulatory requirements for serialization, tracking, and tracing of all pharmaceutical products, including COVID-19 vaccines. It is specifically designed to solve the issues with expected data volumes, queries, and reporting as per the guidelines. SAP ATTP is now playing a critical role in major pharmaceutical companies across the globe.
Leading COVID vaccine producers like Moderna, German vaccine manufacturer CureVac, AstraZeneca, and others are relying on SAP to manufacture and distribute vaccines on a global scale. According to SAP’s report, 18 of the world’s top 20 COVID vaccine producers run their entire product lifecycle and supply chain processes on SAP for better traceability, tracking, and serialization.
Read more: SAP for Business: 9 Ways to Maximize Business Performance
What is the significance of SAP ATTP?
If you have experienced a delay in delivering an urgently needed pharmaceutical product, you can understand the potential impact it can have on people and society. If the first challenge was to create and test vaccines at record speed, the next challenge is to vaccinate people across the globe before the virus mutates and becomes more virulent. This demand is stretching the supply chain to its limits. Digitizing their production to achieve production scale and flexibility is critical as most vaccine producers are data-heavy industries.
What are the advantages of using SAP ATTP software?
SAP ATTP is a solution that facilitates data exchange with packaging lines, supply chain partners and contract manufacturing organizations, and warehouse applications. Here are a few benefits of this software:
- Integrates serialization with business processes
- Enables compliance with directives across markets
- Reduces compliance costs for all serialization requirements
- Streamlines and manages a secure pharmaceutical supply chain
- Packaging line integration
- Boosts efficiency of your supply chain
- Enables simple reporting processes
How can SAP help win the race to vaccinate the world?
For perhaps the first time in history, pharmaceutical companies were pushed to focus all their resources on the development of vaccines in record time. And for the first time in history, they are expected to win the race to vaccinate the global population. This is an opportunity for intelligent enterprises to make a difference. SAP rose to assist by providing various solutions to address this pressing need.
1. Enables collaboration with supply chain partners
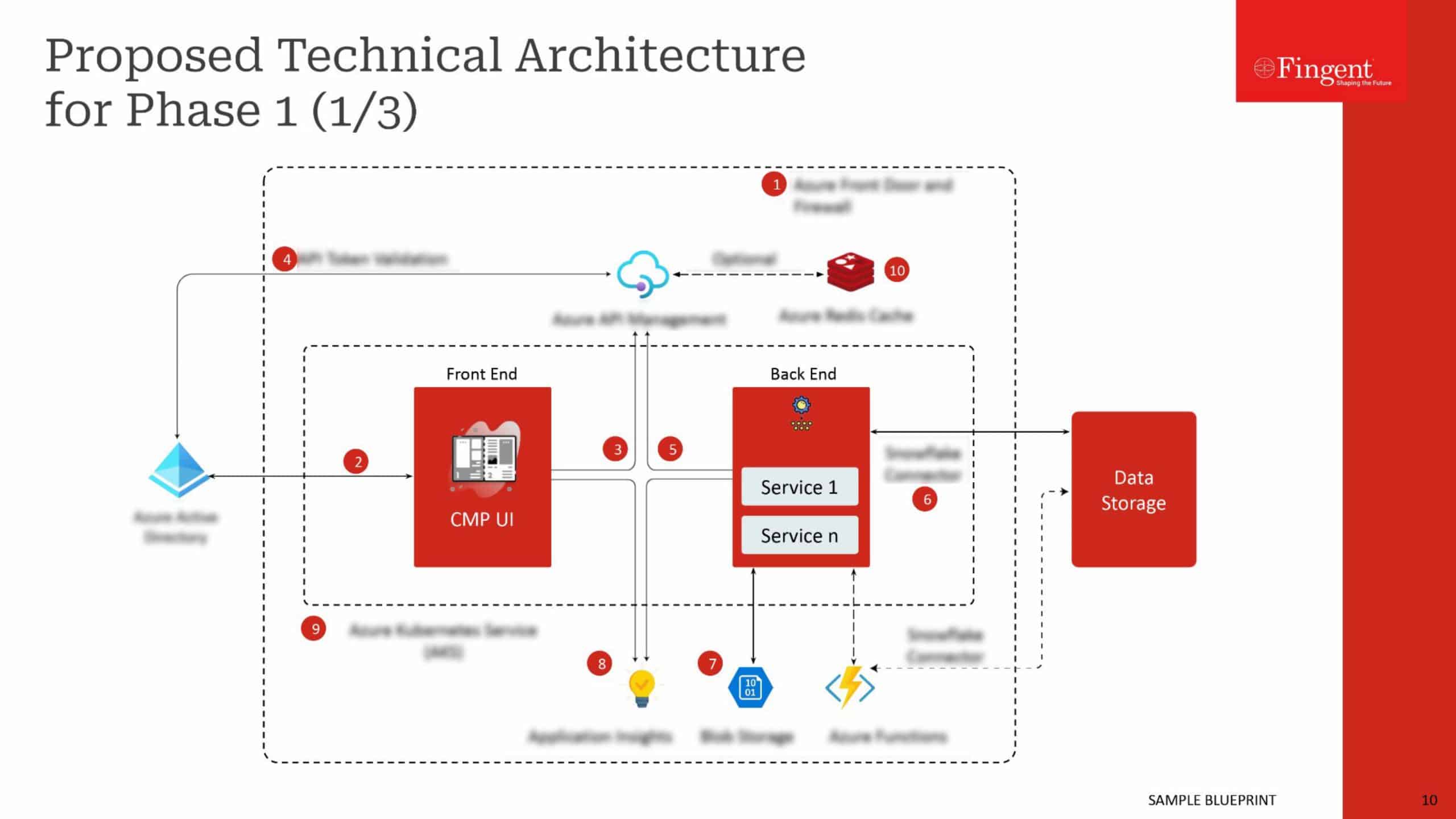
SAP helps pharmaceutical companies comply with international legislation, which focuses on ensuring genuine medicines reach patients and product authentication. SAP applications enable serial number management and regulatory reporting capabilities. Companies can use SAP to facilitate collaboration with supply chain partners, such as contract manufacturers and wholesalers. SAP cloud platform enables trading partners to exchange large amounts of serialization and associated data.
- Pharmaceutical companies must guard against product counterfeiting, reimbursement fraud, diversion, and medication errors.
- A company must include verification in case of the return of vaccine packs or an apparent tampering of the products.
- SAP’s digital network consists of a blockchain-based verification repository to eliminate such counterfeit products along the supply chain.
- Once manufacturers push data into the blockchain, wholesale distributors can execute verification requests. Since these solutions can be integrated into a pharmaceutical company’s backbone, your company enjoys a holistic view of its end-end processes.
Read more: 6 Tips for Logistics and Supply Chain Leaders to Plan COVID-19 Vaccine Distribution Strategies
2. Ensures patient safety and eliminates the risk of fake vaccines
Pharmaceutical thefts have cost billions of dollars worth of losses annually. One measure taken to overcome this challenge was to implement serialization. Serialization of pharmaceuticals protects the end-user and prevents manufacturers from producing fake goods.
SAP serialization allows you to provide a unique identifier (UID). It enables manufacturers to define regulations based on the laws of each country. For example, certain countries may require the entire product dose to be serialized, while others may require each unit to be serialized. SAP allows you to comply according to your country’s regulations. However, serialization is just the first step.
3. Helps you gain visibility of the vaccine from its source to destination
SAP ATTP allows for a transparent journey of vaccine from its source to the destination. Tracking involves knowing the current status, such as the current location of the shipment, in whose possession it is, and the condition of the vaccine, while Tracing involves knowing the past, such as where the vaccine has been, who had it, and was it handled appropriately.
SAP ATTP is beneficial to companies since the early stages of production and distribution. As it allows you to enter all the product details into the system, it is easy to locate where the product is at any given point in the supply chain. Furthermore, SAP can enable withdrawing a specific compromised batch instead of an entire shipment saving a considerable amount of money.
Pharmaceutical regulations vary from country to country. Nevertheless, all pharmaceutical products must be serialized for easy identification within the supply chain. Apart from this, certain governments may require companies to report data to regulatory bodies or authorities. They may also require verification of the products to ensure it is an authentic product. SAP can make these tasks more efficient as it allows you to:
- Generate and format serialized numbers
- Aggregate goods for quick and easy scanning of the vaccine units
Read more: How SAP Helps Realize Voice-enabled Warehouse Operations
Leave no loopholes in the vaccine supply chain
SAP ATTP helps pharmaceutical industries to gain real-time visibility into the location and condition of their product along the entire supply chain. In addition, it helps them perform better management by allowing real-time reporting and analysis to turn insights into actions fueling the mission to vaccinate the world.
As an SAP Silver Partner, Fingent top software development company, can help you implement SAP ATTP seamlessly and cost-effectively. Time is of the essence right now, and vaccine producers need to get the vaccine out faster while ensuring safety and compliance. So let’s save lives together. Connect with our SAP expert to see how you can benefit from implementing Advanced Track and Trace for pharmaceuticals.
Stay up to date on what's new

Recommended Posts

22 Apr 2026 B2B
SAP EWM Implementation: Strategizing a Roadmap to Success
Warehouse operations are no longer predictable environments. Order patterns shift without warning. SKU volumes expand continuously. Fulfillment timelines shrink. Most warehouse systems were not built for this level of volatility.……

24 Feb 2026 B2B
Generative AI with SAP: Transforming Intelligent Enterprises
Enterprises are drowning in data, but still starve for clarity. Not because the data is missing. Because insight does not emerge automatically from systems, even very good ones. This is……

20 Jan 2026 B2B
Why Businesses Must Consider SAP EWM For SAP S/4HANA Cloud Private Edition?
Warehousing has changed radically. Next-day delivery is no longer impressive—it’s assumed. And 95% inventory accuracy? That’s not “good enough” anymore. That’s leakage. As the traditional LE-WM (Logistics Execution – Warehouse……

21 Nov 2025 B2B
Powering Up Procurements with SAP Ariba
Procurement isn’t back‐office drudgery anymore. It’s a strategic powerhouse. It drives savings. It sparks innovation. It can even transform how companies compete. Imagine procurement as your supply‐chain engine. If it’s……
Featured Blogs
Stay up to date on
what's new